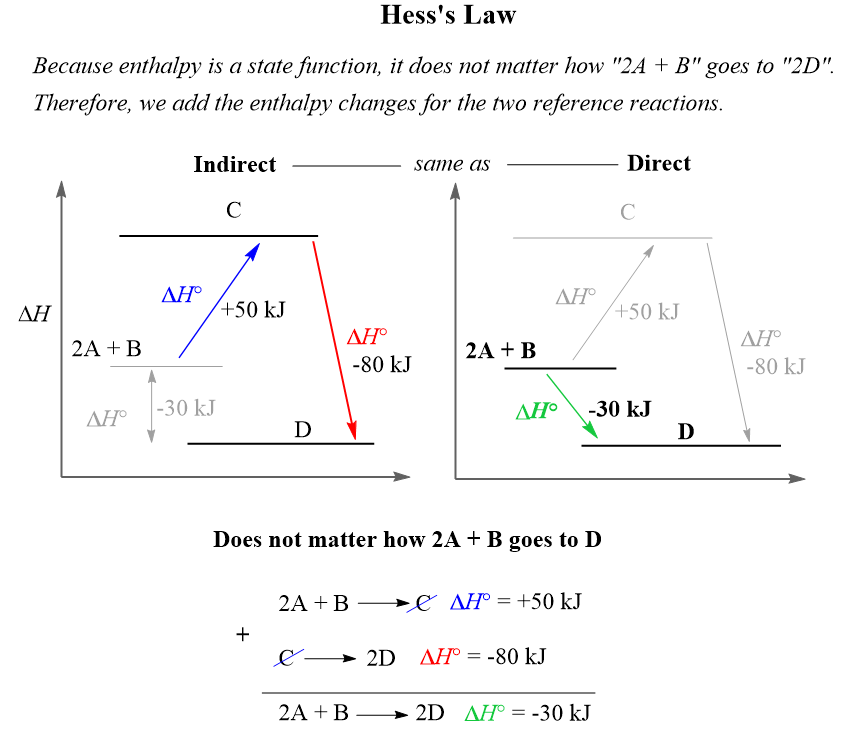
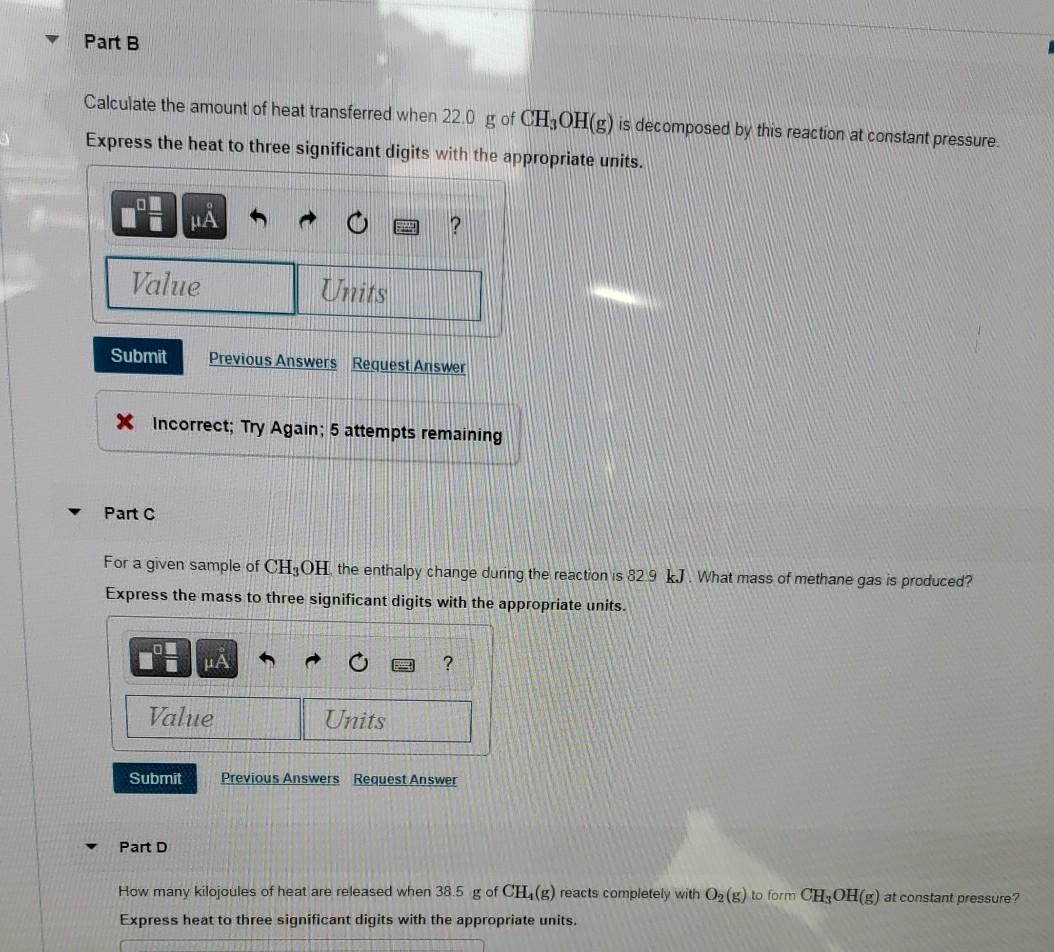
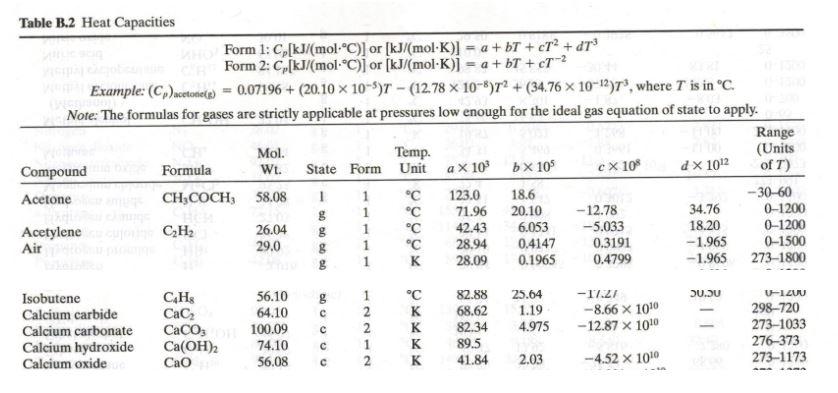
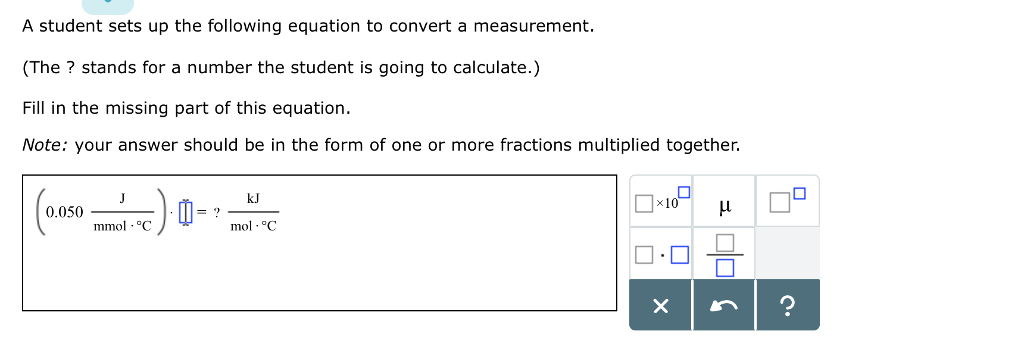
OpenStax College Physics Solution, Chapter 14, Problem 6 (Problems & Exercises) | OpenStax College Physics Answers
![Calculate resonance energy of C(6)H(6) (g). {:("Given :",Delta(f)[C(6)H(6)(g)]=-360 kJ mol^(-1)),(,DeltaH("Sub")[C("graphite")]=716 kJ mol^(-1)),(,B.E.(H-H)=437 kJ mol^(-1)),(,B.E.(C=C)=620 kJ mol^(-1)),(,B.E.(C-C)=340 kJ mol^(-1)),(,B.E.(C-H)=490 kJ ... Calculate resonance energy of C(6)H(6) (g). {:("Given :",Delta(f)[C(6)H(6)(g)]=-360 kJ mol^(-1)),(,DeltaH("Sub")[C("graphite")]=716 kJ mol^(-1)),(,B.E.(H-H)=437 kJ mol^(-1)),(,B.E.(C=C)=620 kJ mol^(-1)),(,B.E.(C-C)=340 kJ mol^(-1)),(,B.E.(C-H)=490 kJ ...](https://d10lpgp6xz60nq.cloudfront.net/web-thumb/32505248_web.png)
Calculate resonance energy of C(6)H(6) (g). {:("Given :",Delta(f)[C(6)H(6)(g)]=-360 kJ mol^(-1)),(,DeltaH("Sub")[C("graphite")]=716 kJ mol^(-1)),(,B.E.(H-H)=437 kJ mol^(-1)),(,B.E.(C=C)=620 kJ mol^(-1)),(,B.E.(C-C)=340 kJ mol^(-1)),(,B.E.(C-H)=490 kJ ...
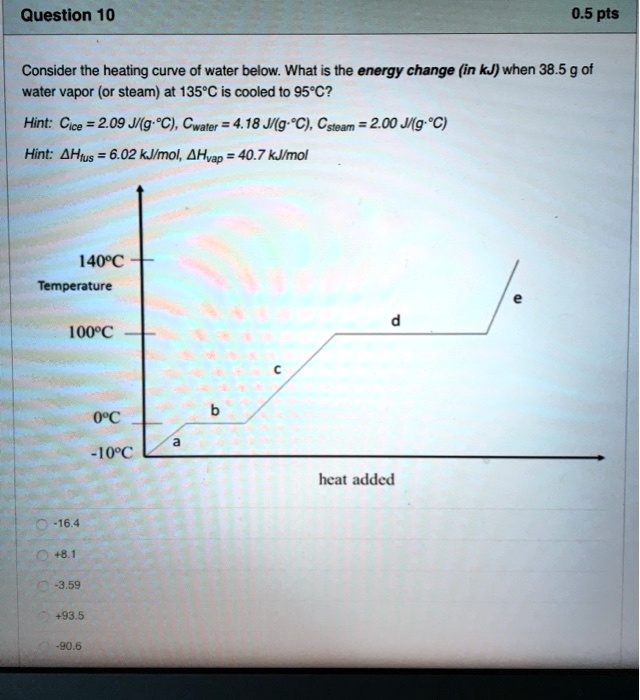
SOLVED: Question 10 0.5 pts Consider the heating curve of water below: What is the energy change (in kJ) when 38.5 9 ot water vapor (or steam) at 135*C is cooled to

What is the temperature change in 224 g of water upon the absorption of 55 kJ of heat, the specific heat of water is 4.18 J/g °C? | Socratic

USB C Tester,KJ-KayJI 2 in 1 Tester Color Screen IPS Digital Multimeter(2022),Voltage,Cur,Pwr,Resistance,Elec,Temp,Capacity,Tme,Fast Charging,with USB Clip Cable Support PD2.0/PD3.0,QC2.0/QC3.0,BC1.2: Amazon.com: Tools & Home Improvement

Calculate the number of KJ of heat necessary to raise the temperature of 60.0g of aluminium from.... - YouTube

The heat of combustion of C(graphite) is - 393.5kJ mol^-1 . The heat of formation of CO2 from graphite is kJ mol^-1 .
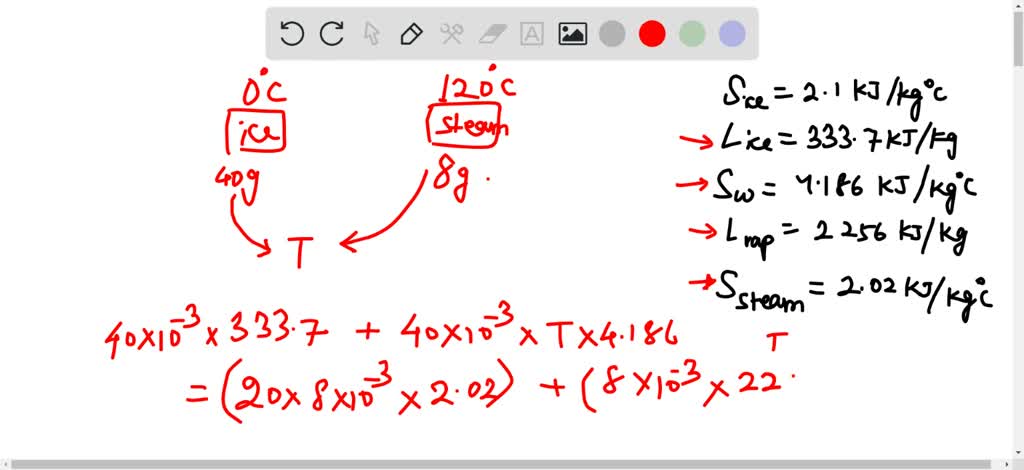
SOLVED: The specific heat of ice is 2.10 kJ/kg C, the heat of fusion for ice at 0 C js 333.7 kJ/kg, the specific heat of water 4.186 kJ/kg C, the heat

A heat pump is used to maintain a house at 25 ? C by extracting heat from the outside air on a day when the outside air temperature is 4 ? C.